Initiatives for innovative container development
IV solution containers that continue to evolve for ease of use and infection prevention

When our company began manufacturing various injectable solutions in 1946, IV solution containers were only made of glass. To overcome the drawbacks of glass containers, which are heavy and prone to breaking, we adopted technology from a Swiss company that was manufacturing plastic bottles for IV solutions, developed Japan’s first plastic bottles for IV solutions, launching them in 1968. While further promoting improvements to increase container transparency and strength, we developed a soft bag for IV solutions in 1986 and since then have successively developed high-performance soft bags such as the double-bag, triple-bag, and quad-bag.
Development of the double-bag, triple-bag, and quad-bag
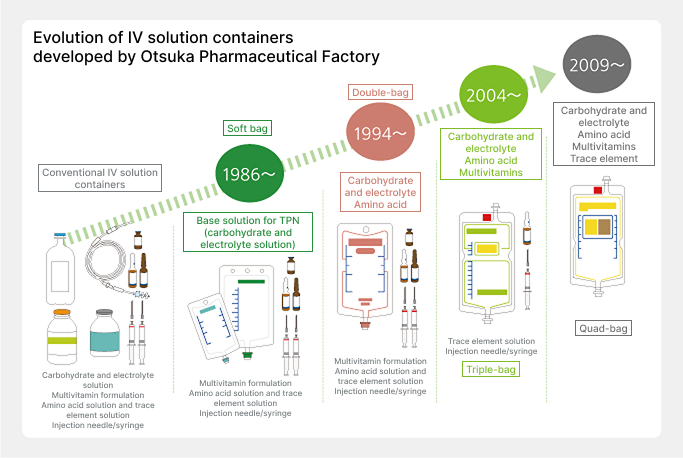
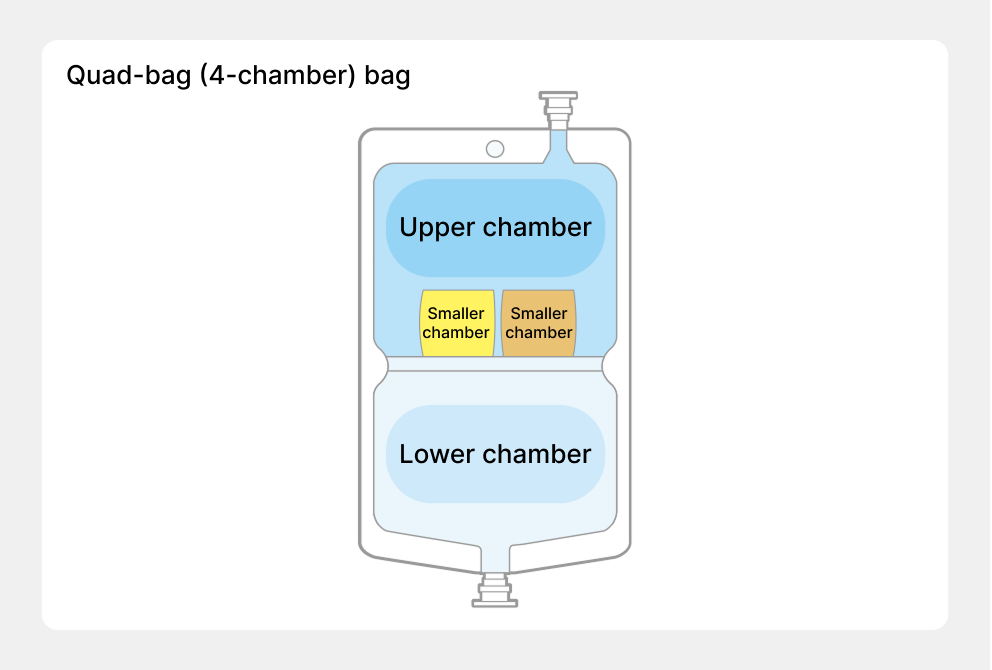
For IV solutions to provide nutrition, it is necessary to mix glucose, amino acids, electrolytes, vitamins, trace elements, and other components. However, when these are mixed and stored for a long time, chemical changes occur causing these components to deteriorate. Therefore, until then, each product had been mixed at medical institutions just before use, but there were risks, such as forgetting to mix the components and bacteria contamination. The solution to these problems was the “multi-bag” we developed, the first of which was the “double-bag,” launched in 1994. By separating the drug solutions into two chambers, upper and lower, and opening the partition before use, mixing and preparation can be performed aseptically and easily. In 2004 we launched the “triple-bag,” which added a smaller chamber to the double-bag, and in 2009 we launched the world’s first “quad-bag,” which added two smaller chambers to the soft bag with upper and lower chambers.
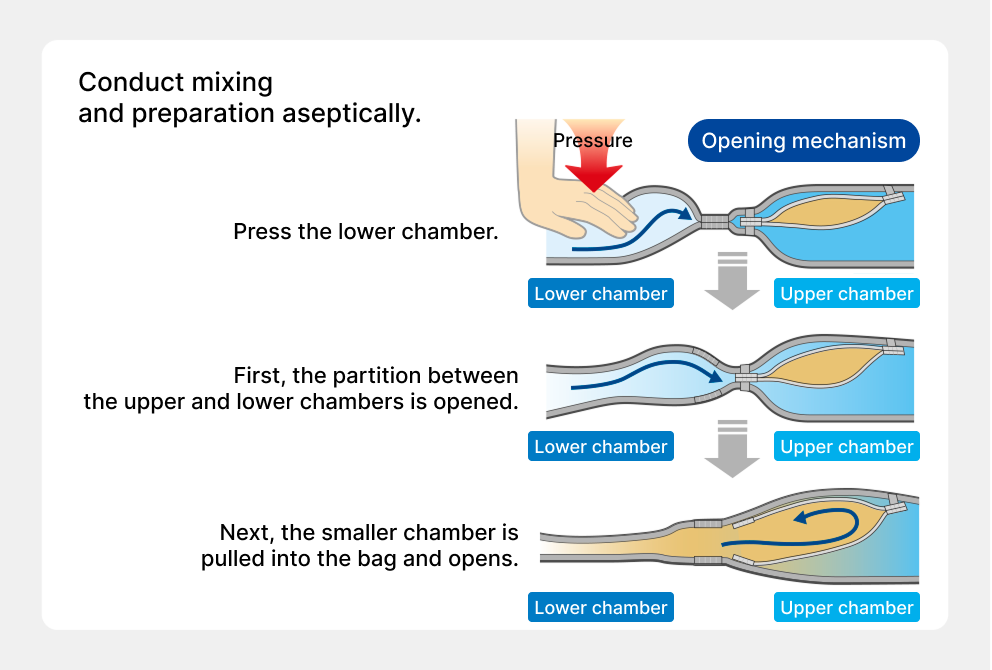
Development of the Otsuka multi-chamber bag system used for antibiotic kit products
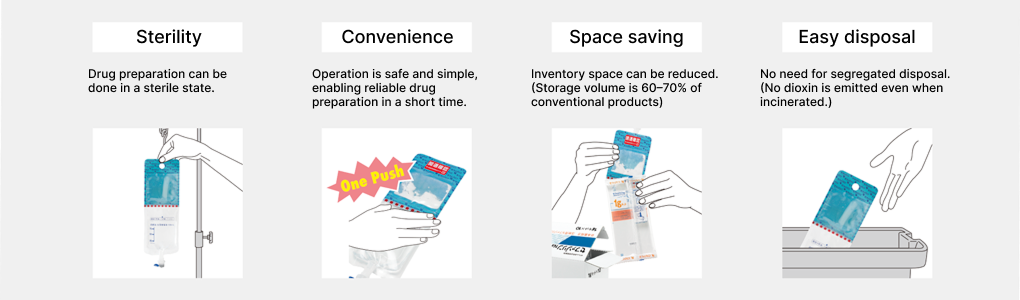
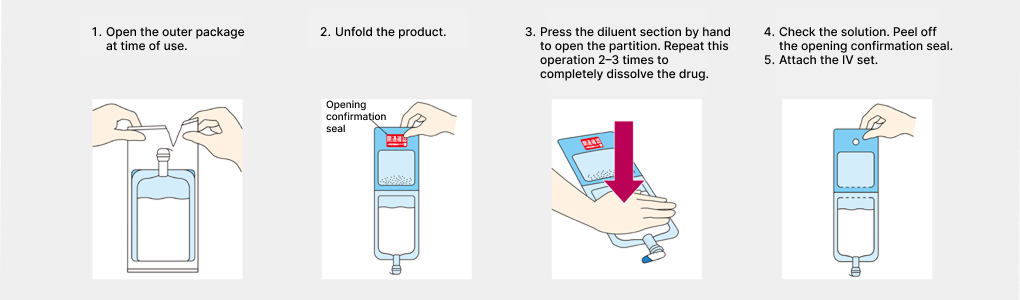
Most injectable antibiotics are in powder form that must be dissolved and prepared. At medical institutions, it has become common to dissolve this in a small-volume diluent of 50–100 mL and administer by drip infusion. Since aseptic handling is required, this posed a significant burden on healthcare workers. We engaged in research and development of containers that allow safer and simpler mixing and preparation, and in 1995 developed the Otsuka multi-chamber bag system, which allows antibiotics and solvents to be administered in a sterile environment with a single press, and developed Japan’s first antibiotic kit product utilizing that technology. As this also helps prevent medical errors such as mix-ups of drug solutions, it has been highly evaluated by medical institutions, and today such systems are also utilized in other companies’ antibiotic kit products.
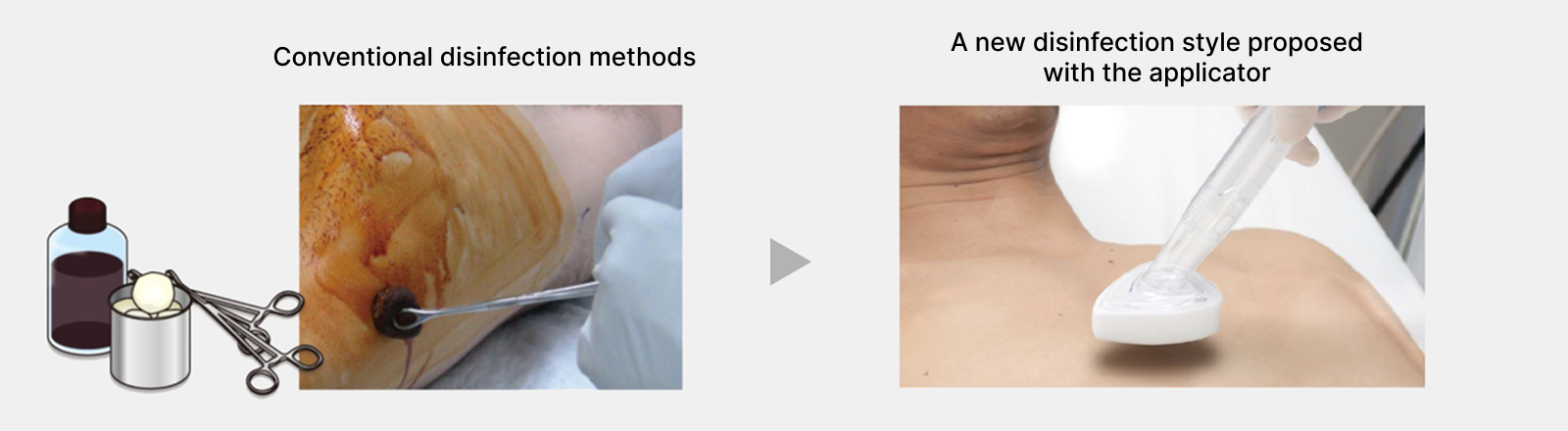
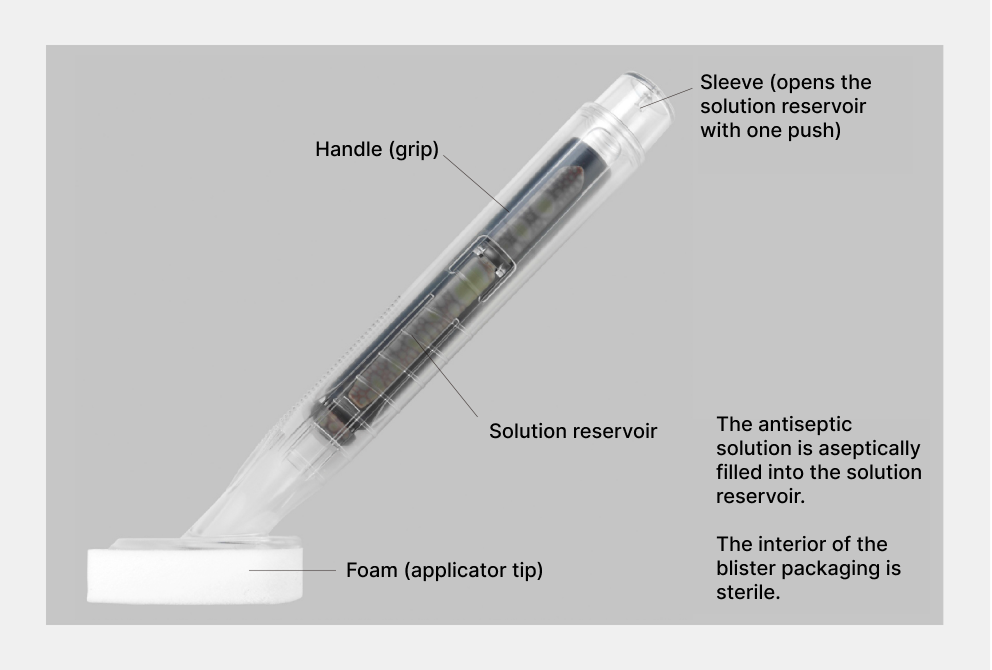
Bringing our proprietary applicator product technology that integrates disinfectant and applicator to Japan
Topical skin antiseptics play a major role in preventing infections during surgery. Our company focused on an active ingredient discovered in 1992 and, after 23 years of persistent research, developed a new topical skin antiseptic, the first in Japan in over 50 years. At the same time, using our proprietary technology, we refined an antiseptic applicator commonly used in U.S. healthcare facilities and launched Japan’s first applicator-type disinfectant that integrates the antiseptic and the applicator. We hope this product will benefit patients and healthcare professionals by enabling simple, hygienic, and rapid application.
Streamlined management and preparation
- Since the antiseptic and applicator are integrated, there is no need to prepare transfer containers, cotton balls, or forceps.
- No need to clean and sterilize instruments.
Hygienic and rapid disinfection
- Since it is a single-use formulation, it is expected to reduce the risk of microbial contamination and foreign matter contamination from reusing solutions.
- With one push, preparation for disinfection is complete, enabling rapid disinfection.