Developing products that benefit patients (Clinical Development Department)

Ideas for new drugs and medical devices that originated at the Naruto Research Institute of the Research and Development Center are provided to the Clinical Development Department, which diligently develops them so they can be turned into reliable products used in medical practice that benefit patients. As the organization involved in clinical trials, the Clinical Development Department is responsible for the final step of product development that has been carefully thought through over many years.
Approach to clinical trials
The Clinical Development Department is responsible for conducting clinical trials with full
ethical consideration, confirming efficacy and safety, and delivering products to patients
as quickly as possible. Specifically, our mission is to “design clinical study plans in line with the development strategy,” “carry out high-quality clinical trials in compliance with laws and regulations,” and “achieve early approval and early market launch.”
To date, we have conducted clinical studies primarily for clinical nutritional products, as well
as for new topical antiseptics/disinfectants and medical devices. A feature of these studies is
that standard evaluation methods are often not established, requiring us to develop clinical evaluation
methodologies from scratch.
So, with what mindset have we conducted clinical studies, how will we conduct them going forward,
and what initiatives will be needed in the future? These are answered by the following.
Clarify and share
the Target Product Profile
Understand the medical setting
Share risks and challenges
We are also working toward the goal of global expansion.
Clarify and share the Target Product Profile
A Target Product Profile (TPP) describes the intended indications/effects, specifications, and
other attributes of the drugs or medical devices under development. By clarifying the TPP, we
can work with a consistent approach from formulation design through preclinical studies to
clinical studies. As a result, the overall team remains aligned, decisions are made more
quickly, and product development can be carried out efficiently.
The scope for sharing the TPP is not limited to internal departments. It extends quite broadly to
include various stakeholders such as regulatory authorities, medical institutions conducting clinical
trials, and CRO (Contract Research Organization). This enables everyone involved in drug development
to collaborate toward the same goal and work in a focused manner, making the process more efficient
and faster.
Understanding the medical setting
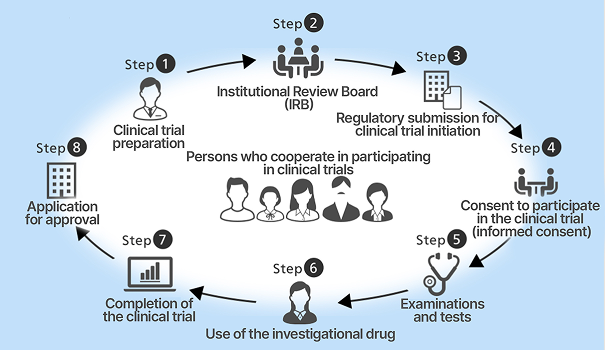
We constantly consider what patients and healthcare professionals truly need and strive to
understand what is happening on the frontline of medicine to meet those needs. Information
we obtain through medical institutions is also shared with the Naruto Research Institute.
Such information is not only important for clarifying the TPP of drugs, medical devices, and
other products under development, but is also leveraged in research on next-generation drugs
and medical devices at the Naruto Research Institute.
Moreover, the development of drugs and medical devices cannot be carried out by a pharmaceutical
company alone without knowing the medical setting. Above all, clinical studies are inconceivable
without actual medical practice, especially without patients. In other words, it is drug development
that leverages the voice of patients (patient centricity).
For example, reflecting the voices of medical professionals on the frontlines and patients in
documents such as clinical study protocols is one way of leveraging patient voices. Doing so helps
protect the human rights of patients, ensure safety, and maintain the quality of clinical studies.
Share risks and challenges
To successfully complete a clinical trial, sharing risks and challenges is absolutely
essential. Before starting clinical studies, thoroughly identifying anticipated risks and
challenges and sharing them among development team members makes it possible to conduct
clinical studies with clear priorities and focus. Here, too, patient-centric drug
development (patient centricity) plays a role.
Meanwhile, while conducting clinical studies, unforeseen events often occur due to unexpected
adverse effects, significant environmental changes, changes in medical technology, and responses
by regulatory authorities. To address such risks, a project management approach that treats clinical
studies as projects becomes important.
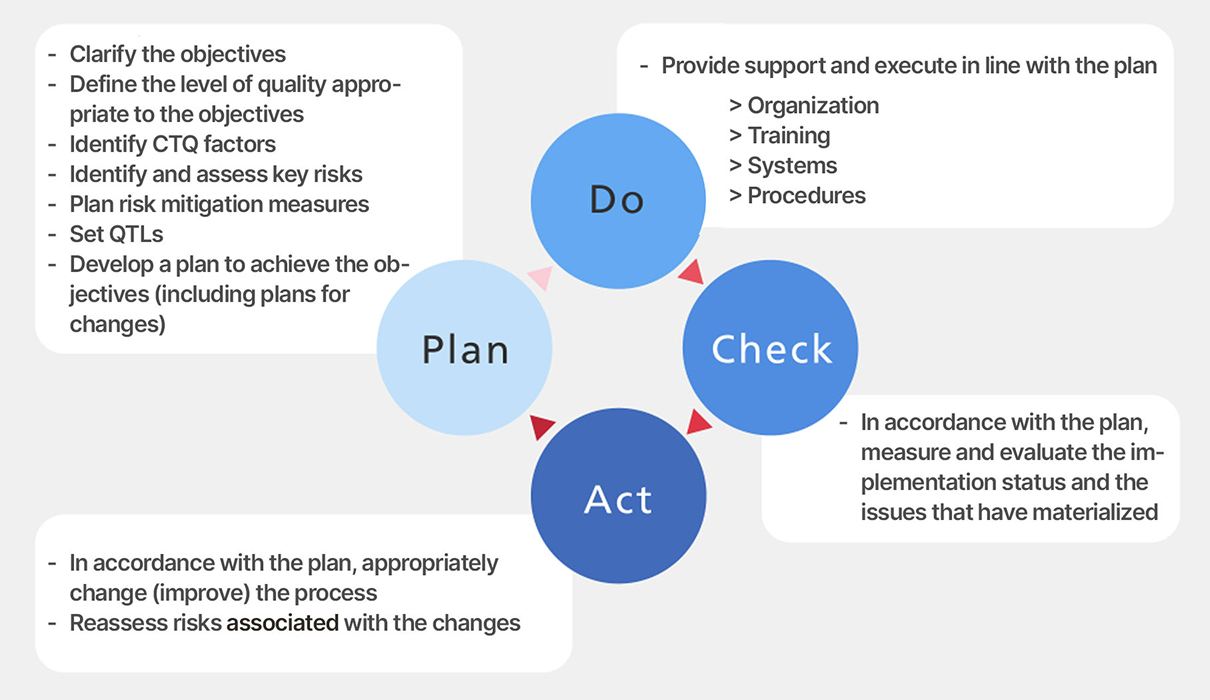
In other words, develop a plan optimized to achieve the objectives (Plan), then, based on the
progress of the clinical studies (the “Do” portion), determine the
extent of deviation between the plan and actual conditions (Check), and proactively take measures
(Action) to address issues.
Expand globally
It goes without saying that healthcare needs have no borders. There are patients around the
world who need drugs and medical devices.
We have worked, in collaboration with related departments such as Regulatory Affairs and the International
Headquarters, to deliver products developed in Japan to Asia, particularly Southeast Asian countries.
Currently, we are also vigorously advancing product development in the United States.
Conducting clinical studies in the United States leads to faster development and approval of new
drugs, enabling more people to access new treatments. As a result, we can improve the health and
well-being of all people.
Furthermore, by experiencing cutting-edge healthcare and innovative clinical studies, our Clinical
Development Department increases its expertise and lays the foundation for conducting clinical studies
with a more global perspective. Partnerships with new stakeholders, starting with global CROs, are
strengthened, allowing us to share knowledge and know-how, which also contributes to building a
stronger organization.



