Compliance
Code of Business Ethics for the Otsuka Group and Our Company
Under the corporate philosophy “Otsuka-people creating new products for better health worldwide,” the Otsuka Group, as a company that contributes to the health of people around the world, acts with a high sense of ethics and with integrity to earn the trust of stakeholders and aims for sustainable growth. To embody that intent, we have established the Otsuka Group Global Code of Business Ethics, setting standards of behavior that everyone involved in our operations should practice. In addition, we have established various global policies (regulations) that further specify the important items defined in the Code, and we work to instill and entrench compliance through regular training and other activities.
- Otsuka Group Global Code of Business Ethics
- Otsuka Group Human Rights Policy
- Otsuka Group Global Anti-Corruption Policy
- Otsuka Group Global Policy for Conflict of Interest
- Otsuka Group Business Partner Code of Ethics
Based on the Global Code of Business Ethics, our company has formulated the Otsuka Pharmaceutical Factory Code of Conduct, which forms the basis of each employee’s behavior and of organizational norms, and promotes compliance. In accordance with these codes of conduct, as a pharmaceutical company that handles products related to people’s health and lives, we aim to meet the expectations of stakeholders such as patients and healthcare professionals and to earn their trust by having each employee act with a high sense of ethics, including legal compliance and bioethics, and with transparency, in line with our corporate philosophy.
Compliance Program
As a concrete guideline for the Otsuka Pharmaceutical Factory Code of Conduct, we have established and implemented the Otsuka Pharmaceutical Factory Compliance Program. Article 1, “Basic Mindset,” of the Compliance Program stipulates the following.
-
Guided by the philosophy “Otsuka-people creating new products for better health worldwide,” we act with the high ethical standards expected while carrying out our corporate activities, including legal compliance and bioethics, to bring it to fruition.
- We aspire to be a sound company and sound businesspeople who are accepted in any country or region around the world, including Japan, and to that end we comply with the laws and rules of each country and act according to the social norms of each region.
- Including cases where we entrust work to Group companies or external contractors involved in research, development, manufacturing, or sales, we strive to fulfill our corporate social responsibility (CSR) through the manufacture and sale of pharmaceuticals and foods and cultural support.
In addition, we have established codes of conduct that employees must observe regarding, among other things, environmental conservation initiatives, prohibition of unfair transactions, protection of personal information, clinical studies, and medical information provision activities.
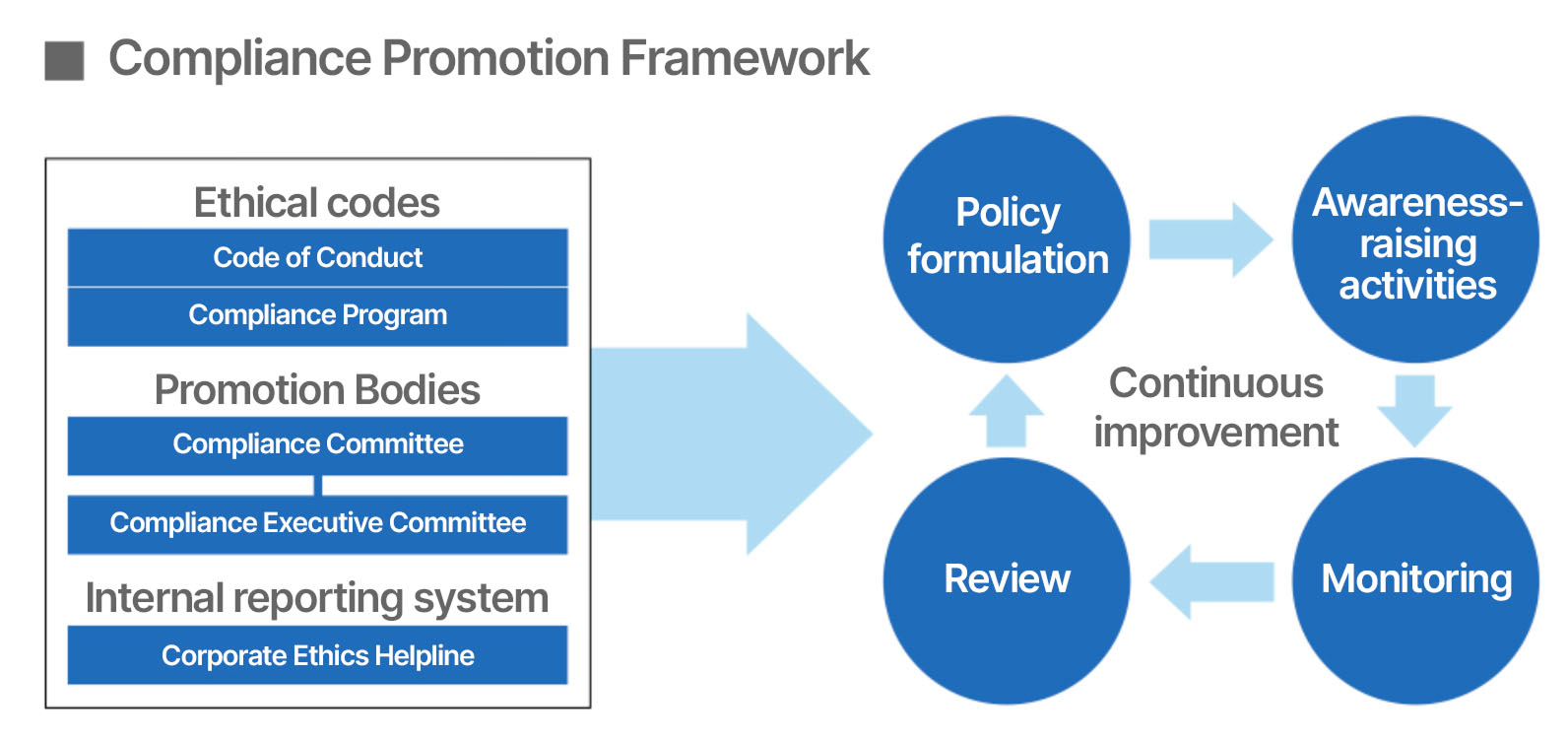
Compliance Promotion Framework
To reliably advance our compliance efforts, we have set out ethical codes such as the
Code of Conduct and the Compliance Program, established promotion bodies such as the
Compliance Committee and the Compliance Executive Committee, and created an internal
reporting system called the Corporate Ethics Helpline, thereby building a compliance
promotion framework.
To embed the ethical codes among employees, we hold forums and study sessions as awareness-raising
activities and monitor the penetration and establishment of corporate ethics through periodic
audits and conduct reviews to drive continuous improvement, thereby ensuring compliance with
relevant laws and the thorough implementation of our corporate philosophy.
Education and Training
We incorporate compliance topics into a variety of training programs, including new employee training and programs for managers. In addition, for directors, compliance executive officers (department heads), and members of the Compliance Executive Committee, we hold annual in-house lectures and forums with external speakers to instill the significance and importance of compliance.
Furthermore, to ensure thorough compliance by each employee, we disseminate information via the intranet and regularly distribute an email newsletter featuring timely topics useful for day-to-day work, and each department conducts small-scale training sessions using these as teaching materials. Through these activities, we are working to develop employees with a strong sense of ethics across the company.
Reporting Hotlines
Internal Reporting Hotline
For compliance, it is important to build a workplace environment where employees feel it is
easy to work and communication is open. We have set up an internal reporting hotline to
prevent and promptly detect incidents arising from legal violations or misconduct and to
enable the organization to function in a self-correcting manner. We accept reports not only
from employees but also from contract and temporary staff, part-time and hourly employees, and
former employees (within one year of leaving). To ensure reporters can use the system without
fear of retaliation, we allow anonymous reporting.
In addition, information about reporting parties and reports is strictly managed as confidential
and, taking into account the need for appropriate investigations and corrective actions, is not
shared with anyone other than those who need to know so that reporters do not suffer any unfair
treatment.
Speak Up Line for Business Partners
Business partners with a business relationship with us may use the Reporting Hotline for Business Partners operated by Otsuka Holdings Co., Ltd.
Speak Up Line for Business Partners- This reporting channel is a web-based hotline that enables business partners to anonymously report fraud and misconduct in their transactional and contractual relationships with companies across the Otsuka Group. Otsuka Holdings Co., Ltd. uses NAVEX’s EthicsPoint system.
Code of Practice
The Japan Pharmaceutical Manufacturers Association has established the JPMA Code of
Practice to ensure greater ethical standards and transparency among its member companies
and to enhance trust in the pharmaceutical industry as a whole. It covers interactions
between all officers and employees of member companies and researchers, healthcare
professionals, patient organizations, and others.
Although we are not a member company of the Japan Pharmaceutical Manufacturers Association,
as a member of the IV Solutions Council, an industry-specific body under the Federation of Pharmaceutical
Manufacturers’ Associations of Japan, which also includes the Japan Pharmaceutical Manufacturers
Association, we have adopted the JPMA Code of Practice and established the Otsuka Pharmaceutical
Factory Code of Practice, which evolves the Ethical Drugs Promotion Code, a code of conduct
that previously applied to the Sales Division. Going forward, we will continue to strive for
appropriate collaboration with healthcare professionals based on relationships of trust. We
comply with laws and corporate ethics to fulfill our responsibilities as a pharmaceutical company
so that appropriate medical care is provided from the patient’s perspective.

Transparency Guidelines
To enhance the transparency of the relationships between our corporate activities and medical institutions and healthcare professionals, we have established the Guidelines on Transparency of Relationships between Corporate Activities and Medical Institutions, etc. and disclose information accordingly.
