Otsuka Pharmaceutical Factory launches Lilium one, a medical device that measures urine volume in the bladder specialized for single measurement
Otsuka Pharmaceutical Factory, Inc. (Head Office: Naruto, Tokushima, Japan; President and Representative Director: Shinichi Ogasawara; "OPF") will launch a portable ultrasonic diagnostic device for the bladder "Lilium one®" on March 1, 2023.
 This product is a medical device that measures urine volume in the bladder using ultrasound. While "Lilium α-200" and its successor "Lilium IP200" are mainly designed for continuous measurement of urine volume, this product was developed to meet the needs of medical professionals who want to measure urinary bladder volume more conveniently and whenever necessary, and is specialized for single measurement. The product was named "Lilium one" because the part in contact with the patient (the probe) and the main unit are integrated into one unit, and measurement can be performed with a single push of a button with one hand.
This product is a medical device that measures urine volume in the bladder using ultrasound. While "Lilium α-200" and its successor "Lilium IP200" are mainly designed for continuous measurement of urine volume, this product was developed to meet the needs of medical professionals who want to measure urinary bladder volume more conveniently and whenever necessary, and is specialized for single measurement. The product was named "Lilium one" because the part in contact with the patient (the probe) and the main unit are integrated into one unit, and measurement can be performed with a single push of a button with one hand.
There is a growing need for bladder function checks (checking and understanding patterns of residual urine volume, urine storage, and voiding volume) and urinary care for patients with frequent urination, urine leakage, and difficulty passing urine due to various diseases. In the medical and nursing care fields in Japan, which has entered a super-aging society, the importance of urinary independence has been recognized, as evidenced by the expansion of the urinary independence guidance fee, which was newly established in the 2008 revision of medical service fees and renamed the additional fee for urinary independence support in the 2020 revision of medical service fees. In addition, residual urine volume is used as an indicator for diagnosis and treatment of dysuria, and residual urine measurement has become an essential test in medical guidelines related to dysuria.
The product will be marketed by OPF under an exclusive sales agreement with the manufacturer and distributor, Lilium Otsuka Co., Ltd. (Head Office: Sagamihara, Kanagawa, Japan; President and Representative Director: Isao Shirasaki; "Lilium Otsuka"). Lilium Otsuka, which has sold medical devices that use ultrasound since 2015, has developed this product by leveraging its expertise in medical device development and manufacturing.
The management vision of OPF is to be "The Best Partner in Clinical Nutrition." In addition, we are expanding our business areas to cover the entire healthcare process, such as prevention, diagnosis, treatment, and monitoring. Among these, we have been enhancing our products in the fields of urology and urination, including the marketing of Lilium α-200 since 2016, and launching its successor product, Lilium IP200, this year, as well as intermittent urinary catheters, Actreen and OT-Balloon Catheter. We will continue to develop products that can contribute to excretion care for patients.
Based on the corporate philosophy of "Otsuka-people creating new products for better health worldwide," the Otsuka Group is dedicated to contributing to the health of people around the world.
Product Information
| Generic name | Portable Ultrasonic Diagnostic Device for the Bladder |
| Product name | Lilium one |
| Medical device certification number | 303ADBZX00050000 |
| Accuracy | ±(15% + 20 mL) |
| Measurement result display range |
0 to 600 mL If the measurement result is larger than 600 mL, "> 600 mL" will be displayed. |
| Power source | Rechargeable battery (built-in lithium-ion battery) |
| Class category | Controlled medical device (Class II), Specified controlled medical device |
| Weight | Approx. 200 g |
| Size (mm) | 77.9 (W) × 24.4 (D) × 124.0 (H) |
| Medical fee item | D216-2 Residual urine measurement test |
| Certification date | May 28, 2021 |
| Basic function | Measurement of urine volume in the bladder |
| Principle of measurement | Ultrasonic waves are directed to the bladder, the amount of urine in the bladder is estimated, and displayed numerically. |
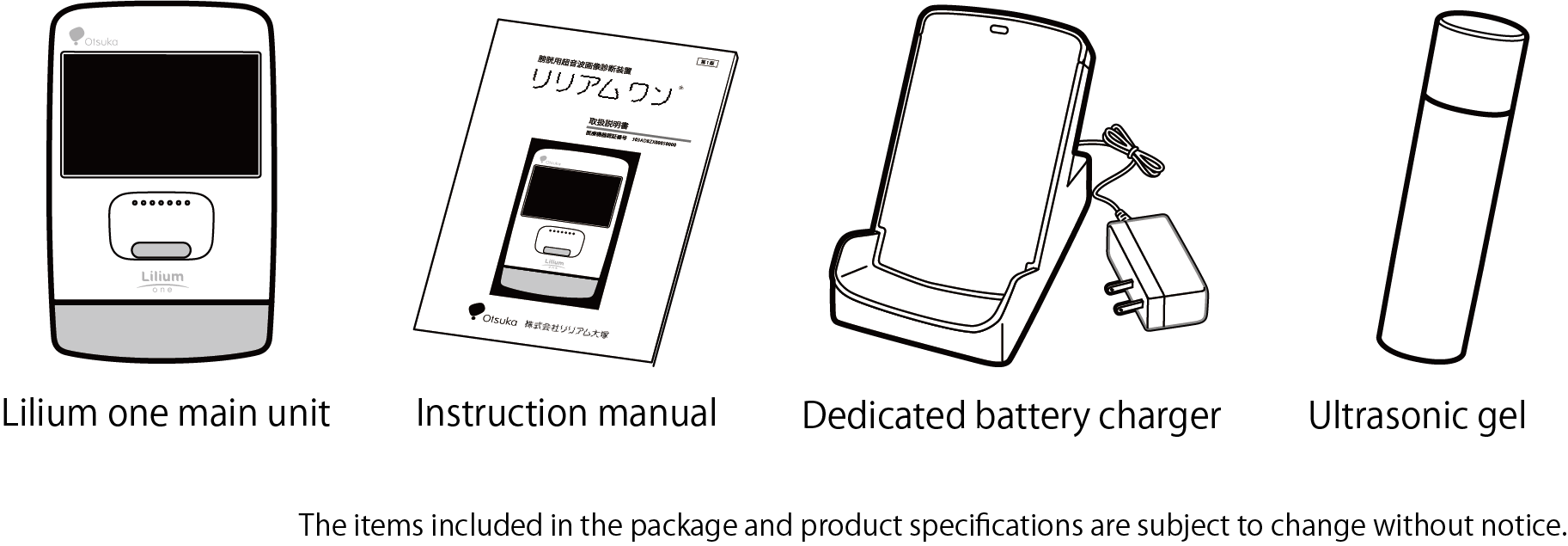
| Configuration |
1) "Lilium one" main unit Probe: Elastomer, flame-retardant ABS 2) Dedicated battery charger for "Lilium one" |
| Suggested retail price | JPY 410,000 (excluding tax) as the Lilium one kit (including the main unit, dedicated battery charger, and ultrasonic gel) |
| Device photo |


|
| Contents of Lilium one kit |
|
